GRS News
Funding competition Fast Start Innovation
12/7/2022
Innovate UK is offering small and micro businesses a share of up to £30 million in grants alongside tailored business support delivered by Innovate UK EDGE. You can apply for up to £50,000 for highly innovative ideas that have a clear route to commercialisation through business growth.
Quick Update: Application Opens 11th July 2022 -- Application Closes 26th July 2022 11:00 am - We are supporting Healthcare innovations with Regulatory Strategy.
Your project must lead to new products, processes or services that are significantly ahead of others currently available, or propose an innovative use of existing products, processes or services. It can also involve a new or innovative business model.
In your application, you must be able to show that you:
- have a great idea
- need public funding
- have the capability to deliver the project
- will deliver the project
Your idea must focus on either achieving net zero or self-driven healthcare, and how new technology can enable affordable, adoptable and investable innovations in these areas. We are particularly interested in proposals which will help you:
- develop both your idea and your business
- create a new revenue stream
- evidence and enable your market, funding and commercialisation plans
- show what’s feasible, helping you decide whether to pursue your idea further
- catalyse further innovation on your path to commercial success
- respond to changing market conditions
Innovate UK runs many different competitions, and each will have different eligibility and scope requirements. The guidance in this competition brief will always supersede any other guidance. If you are unsure or have any questions, please contact our Customer Support Service who will assist you further.
In applying to this competition, you are entering into a competitive process. This competition closes at 11am UK time on the deadline stated.
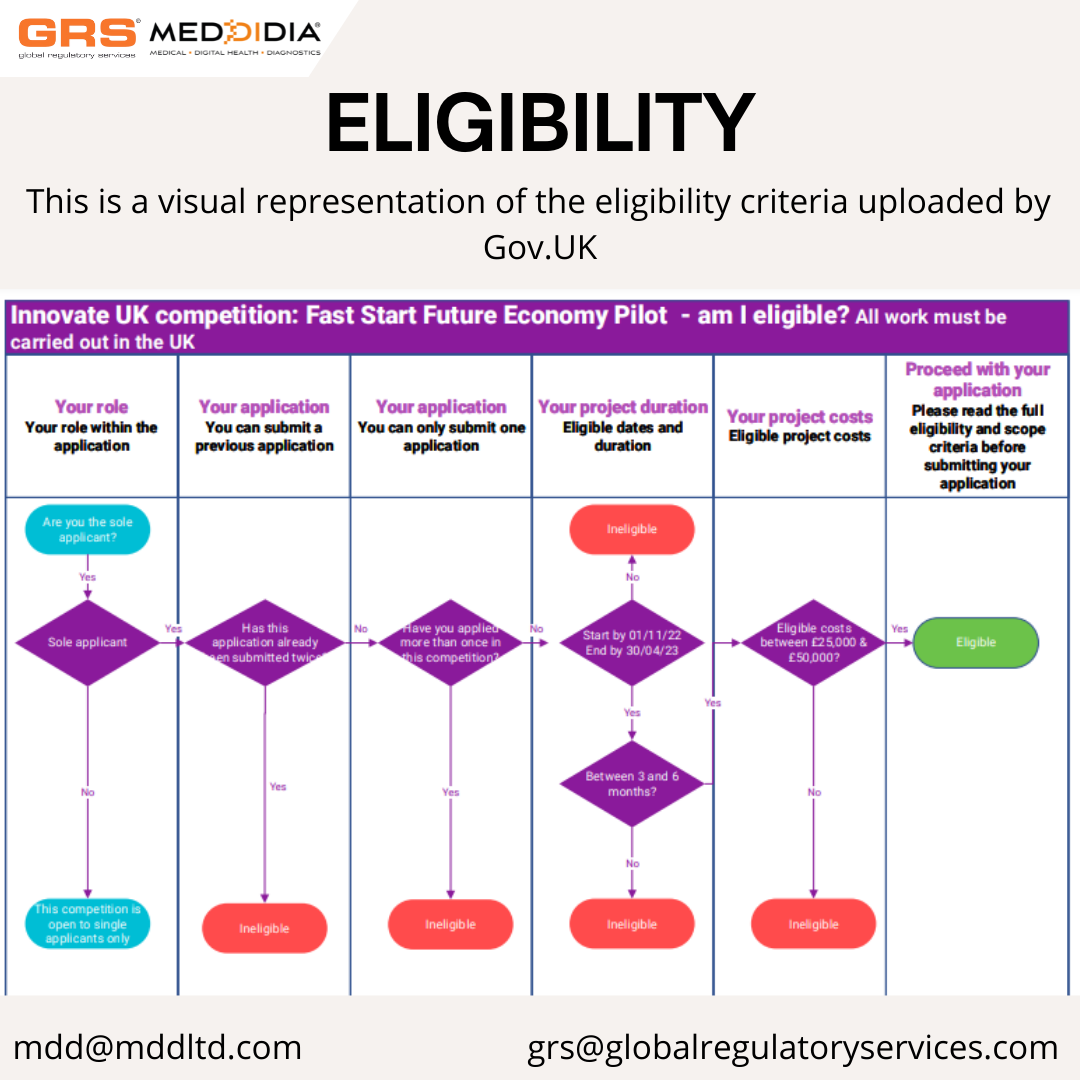
This grant is for projects whose total costs must be between £25,000 and £50,000.
Who can apply
Your project must:
- have total project costs between £25,000 and £50,000
- start by 1 November 2022
- end by 30 April 2023
- last between 3 and 6 months in duration
- carry out its project work in the UK
- intend to exploit the results from or in the UK
You must only include eligible project costs in your application. If you exceed the total project costs of £50,000 you will be ineligible and your application will not be sent for assessment.
Under current restrictions, this competition will not fund any procurement, commercial, business development or supply chain activity with any Russian entity as lead or subcontractor. This includes any goods or services originating from a Russian source.
You will be made ineligible if you exceed the Minimal Financial Assistance limit. You must submit a completed declaration as part of your application.
Lead organisation
To lead a project your organisation must:
- be a UK registered micro or small business
- not have previously been awarded funding from Innovate UK (recipients of Young Innovators and Women In Innovation awards are exempt from this and are eligible to apply)
- not have exceeded your £315,000 limit of de minimis or minimal financial assistance funding during the current and previous 2 fiscal years
Subcontractors
Subcontractors are allowed in this competition.
Subcontractors can be from anywhere in the UK and you must select them through your usual procurement process.
You can use subcontractors from overseas but must make the case in your application as to why you could not use suppliers from the UK.
You must provide a detailed rationale, evidence of the potential UK contractors you approached and the reasons why they were unable to work with you. We will not accept a cheaper cost as a sufficient reason to use an overseas subcontractor.
All subcontractor costs must be justified and appropriate to the total project costs.
Number of applications
You can only apply into this competition with 1 application. We will only award grant funding to 1 project per business.
You can be a subcontractor in any number of applications.
Challenge themes
Your project must focus on how one or more of the seven technology areas outlined here, can enable affordable, adoptable, and investible solutions for either:
Achieving net zero and reducing environmental impact or
Self-driven healthcare, de-centralised healthcare and mental wellbeing through:
- prevention and self-management of lifestyle diseases using personalised, intuitive, consumer-focused and accessible innovations
- maximising the utility, safety and security of personal health records, including self-generated data
- assessing new business models and routes to market for products and services related to self-care and self-management
- developing innovations that support self-management of mental wellbeing
But Why is this information on our website?
Our Experts at Global Regulatory Services [GRS] Ltd. and
Med-Di-Dia Ltd. are helping healthcare innovators in getting their ideas to the market.
Now, in order to bring a healthcare product to the market it should:
- Comply With Regulatory Requirements
- Have a Quality Management System in place
- If selling internationally, the product should comply with international regulations
If the regulatory requirements are not met, then, the innovation is only an innovation!
It cannot be on the shelves!
In order to eliminate the risk of Regulatory Non-Compliance, Our Experts at Global Regulatory Services [GRS] Ltd. and Med-Di-Dia Ltd. will help you with
Regulatory Strategy Report for Market Access and Commercialisation
While developing your Healthcare innovation and the sales plan, you will spend time determining and making careful decisions about your Target Market. The commercialisation strategy and plan should also consider the regulatory requirements of your target markets. Too often, manufacturers will ignore the regulatory requirements in their target market negatively impacting on the market reach of their innovation.
Lucrative factors like revenue and market share often overshadow the regulatory requirements, and manufacturers are lured into spending more time on economic advantage and, at their peril, ignore the regulatory considerations. While deciding on the target market, manufacturers should weigh up the potential lucrative economic factors with the regulatory requirements of that target market.
A well-thought-out regulatory strategy will provide you with a pathway to enhance your production, business development and marketing plans. Shortcomings in the regulatory strategy will directly impact the product's reach in the market and hamper business activities. To avoid potential catastrophe, focus on this 3 point formula:
- Describe your product and check the Regulatory requirements against this description
- Plan your market access activities in parallel to your regulatory strategy (Regulatory and Marketing strategy can go hand in hand)
Get the Complete Advantage of Meeting the Regulatory Requirements for your HEALTHCARE Innovation!
Drop us an email at grs@globalregulatoryservice.com
Get More details on the Fund by clocking here - Details of the Fund on New Page