GRS News
Global Regulatory Services and HealthTech Enterprise Announce Strategic Collaboration to Expand Support for HealthTech Innovators
7/8/2025
We are proud to announce a strategic collaboration between Global Regulatory Services (GRS) and HealthTech Enterprise (HTE). This collaboration unites two trusted organisations with a shared commitment to accelerating innovation, improving patient outcomes, and enabling the successful commercialisation of cutting-edge health technologies.
This partnership creates a powerful platform for end-to-end support across the entire health tech lifecycle—from product development and regulatory compliance to health technology assessment, market access, and post-market adoption strategies.
By combining GRS’s global regulatory and quality compliance expertise with HTE’s deep NHS engagement and market access capabilities, we offer an expanded portfolio of services tailored to the unique needs of early-stage innovators, scaling businesses, and international medtech companies entering the UK and EU healthcare markets.
What This Collaboration Offers:
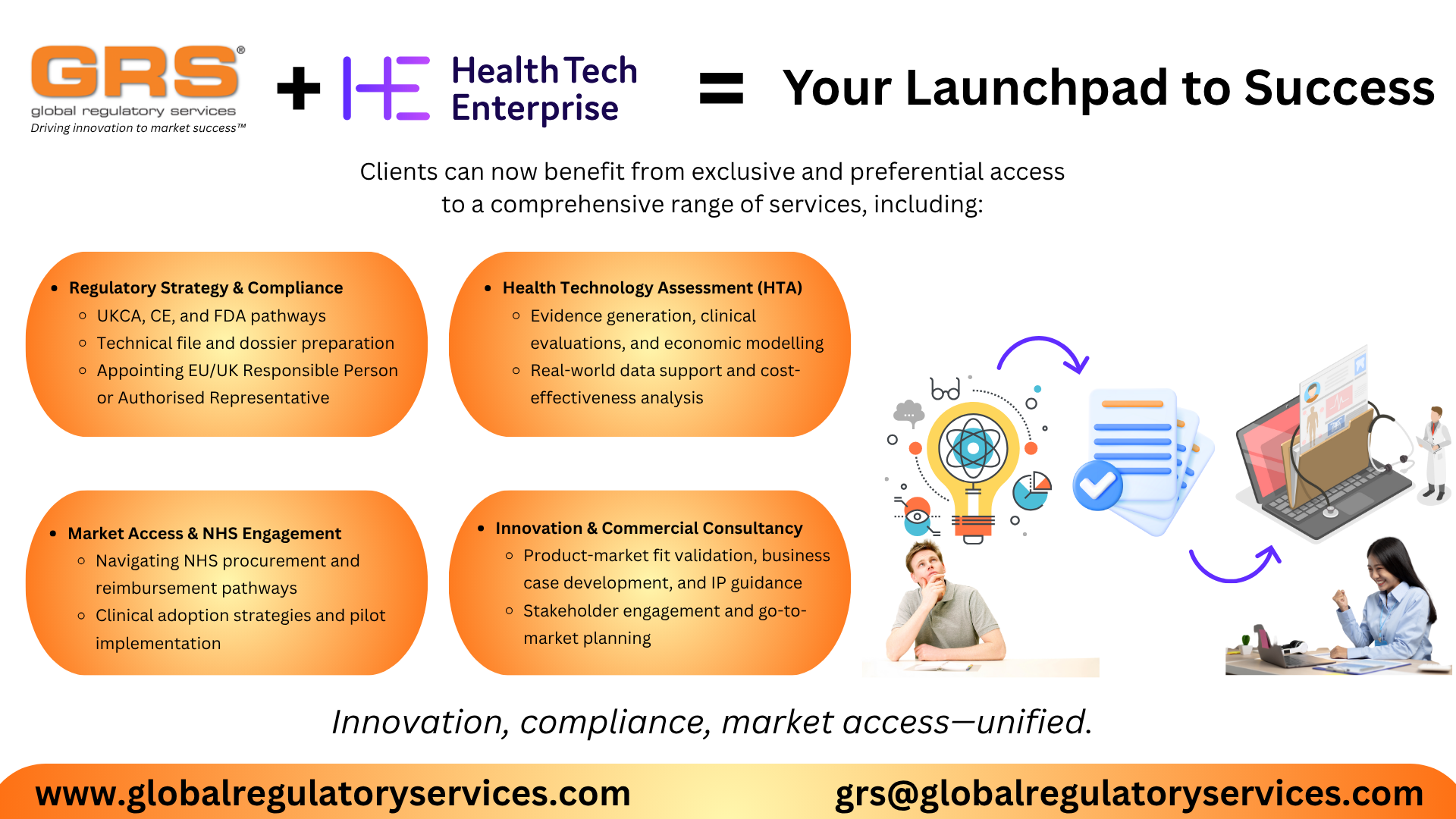
Clients can now benefit from exclusive and preferential access to a comprehensive range of services, including:
- Regulatory Strategy & Compliance
- UKCA, CE, and FDA pathways
- Technical file and dossier preparation
- Appointing EU/UK Responsible Person or Authorised Representative
- Health Technology Assessment (HTA)
- Evidence generation, clinical evaluations, and economic modelling
- Real-world data support and cost-effectiveness analysis
- Market Access & NHS Engagement
- Navigating NHS procurement and reimbursement pathways
- Clinical adoption strategies and pilot implementation
- Innovation & Commercial Consultancy
- Product-market fit validation, business case development, and IP guidance
- Stakeholder engagement and go-to-market planning
This collaboration strengthens our ability to guide healthtech innovators through the increasingly complex global landscape, offering actionable, expert-driven support at every stage of development and commercialisation.
Whether you're preparing to launch a novel medical device, navigating regulatory hurdles, or seeking to establish a foothold within the NHS, GRS and HTE are here to help streamline your journey with transparent, credible, and commercially focused solutions.
Get in Touch
For more information on how this partnership can support your healthtech innovation, please get in touch with us at grs@globalregulatoryservices.com or submit your enquiry via our contact form at: https://globalregulatoryservices.com/global-regulatory-services-contact-details
For more on this article or to enquire about the support available please contact HTE on enquiries@healthtechenterprise.co.uk
We look forward to helping you bring your innovation to life—faster, smarter, and with confidence.